MDC1 (基因名), Mediator of DNA damage checkpoint protein 1 (蛋白名), MDC1_HUMAN.
产品名称:
Human MDC1/ Mediator of DNA damage checkpoint protein 1 ELISA Kit
货号:
-
商标:
EIAab®
监管等级:
别名:
Nuclear factor with BRCT domains 1, KIAA0170, NFBD1
检测方法:
ELISA
特异性:
Natural and recombinant human Mediator of DNA damage checkpoint protein 1
样品类型:
Serum, plasma, tissue homogenates, cell culture supernates and other biological fluids
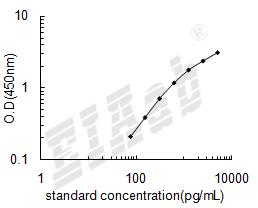
样品数据:
登录.
研究领域:
-
通用注释
亚单元:
Homodimer. Interacts with several proteins involved in the DNA damage response, although not all these interactions may be direct. Interacts with H2AFX, which requires phosphorylation of H2AFX on 'Ser-139'. Interacts with the MRN complex, composed of MRE11, RAD50, and NBN. Interacts with CHEK2, which requires ATM-mediated phosphorylation of 'Thr-68' within the FHA domain of CHEK2. Interacts constitutively with the BRCA1-BARD1 complex, SMC1A and TP53BP1. Interacts with ATM and FANCD2, and these interactions are reduced upon DNA damage. Also interacts with the PRKDC complex, composed of XRCC6/KU70, XRCC5/KU80 and PRKDC/XRCC7. This interaction may be required for PRKDC autophosphorylation, which is essential for DNA double strand break (DSB) repair. When phosphorylated by ATM, interacts with RNF8 (via FHA domain). Interacts with CEP164. When phosphorylated, interacts with APTX (via FHA-like domain).
功能:
Required for checkpoint mediated cell cycle arrest in response to DNA damage within both the S phase and G2/M phases of the cell cycle. May serve as a scaffold for the recruitment of DNA repair and signal transduction proteins to discrete foci of DNA damage marked by 'Ser-139' phosphorylation of histone H2AFX. Also required for downstream events subsequent to the recruitment of these proteins. These include phosphorylation and activation of the ATM, CHEK1 and CHEK2 kinases, and stabilization of TP53 and apoptosis. ATM and CHEK2 may also be activated independently by a parallel pathway mediated by TP53BP1.
亚细胞位置:
Nucleus
Chromosome
Associated with chromatin. Relocalizes to discrete nuclear foci following DNA damage, this requires 'Ser-139' phosphorylation of H2AFX. Colocalizes with APTX at sites of DNA double-strand breaks.
数据库链接
您可能感兴趣
该产品尚未在任何出版物中被引用。
[1].
人MDC1ELISA试剂盒可以做多少个样本?
人MDC1ELISA试剂盒分为2种规格,96孔和48孔。96孔的试剂盒,标曲和样本都做复孔的话,可以检测40个样本。96孔的试剂盒,标曲和样本都不做复孔的话,可以检测88个样本。
[2].
人MDC1ELISA试剂盒使用视频?
人MDC1ELISA试剂盒实验操作视频在以下网址中,对每一步的实验步骤都做了演示,方便实验员能更好地理解ELISA实验的过程。
https://www.eiaab.com.cn/lesson-tech/805.html
https://www.eiaab.com.cn/lesson-tech/805.html
[3].
人MDC1ELISA试剂盒是放在-20℃冰箱保存吗?
EIAab的人MDC1ELISA试剂盒,洗涤液、底物、终止液保存于4℃,其余试剂-20℃冰箱保存。
[4].
人MDC1ELISA试剂盒原理?
双抗体夹心法:用纯化的抗体包被微孔板,制成固相抗体,往包被有固相抗体的微孔中依次加入标准品或受检样本、生物素化抗体、HRP标记的亲和素,经过彻底洗涤后用底物TMB显色。用酶标仪在450nm波长下测定吸光度(OD值),计算样本浓度。
竞争法:用纯化的抗体包被微孔板,制成固相抗体,往包被有固相抗体的微孔中依次加入标准品或受检样本和生物素标记的目标分析物,受检标本中抗原与生物素标记抗原竞争结合有限的抗体。再加入HRP标记的亲和素,经过彻底洗涤后用底物TMB显色。用酶标仪在450nm波长下测定吸光度(OD值),计算样本浓度。
竞争法:用纯化的抗体包被微孔板,制成固相抗体,往包被有固相抗体的微孔中依次加入标准品或受检样本和生物素标记的目标分析物,受检标本中抗原与生物素标记抗原竞争结合有限的抗体。再加入HRP标记的亲和素,经过彻底洗涤后用底物TMB显色。用酶标仪在450nm波长下测定吸光度(OD值),计算样本浓度。
[5].
人MDC1ELISA试剂盒中需要使用的样品量是多少?
夹心法100μL/孔,竞争法50μL/孔。如样本浓度过高时,应对样本进行稀释,以使稀释后的样本符合试剂盒的检测范围,计算时再乘以相应的稀释倍数。
[6].
如何分析人MDC1ELISA试剂盒数据?
建议标准曲线,并计算样本浓度。对于elisa的曲线拟合,一般建议采用4参数曲线拟合,4参数曲线拟合通常更适合免疫分析。推荐使用专业软件进行曲线拟合,例如curve expert 1.3。根据样本的OD值由标曲查出相应的浓度,再乘以稀释倍数;或用标准物的浓度与OD值计算出标曲的回归方程式,将样本的OD值代入方程式,计算出样本浓度,再乘以稀释倍数,即为样本的实际浓度。以下链接是curve expert 1.3软件拟合曲线的方法。
https://www.eiaab.com.cn/news/502/
https://www.eiaab.com.cn/news/502/
[7].
人MDC1ELISA试剂盒中是否包含人和动物的副产物,是否包含感染的或者传染性原料如HIV等?
除了抗体和稀释液中的BSA,不含其它人和动物的副产物,也不含感染材料。
[8].
收集人MDC1ELISA试剂盒血浆样本,用什么作为抗凝剂?
一般建议用EDTA和肝素作为抗凝剂。
[9].
人MDC1ELISA试剂盒酶标板可以拆成几部分?拆的时候是否需要避光,无菌?
人MDC1ELISA试剂盒酶标板是8×12孔条,可拆卸,板子可以拆成12条,注意避免孔污染,不需要避光和无菌。暂时不用的板子,放回原来装的袋子里,密封保存。
[10].
人MDC1ELISA试剂盒样本如何保存?
尽量检测新鲜样本。若无新鲜样本,则4℃保存1周,-20℃保存1个月,-80℃保存2个月。
反馈墙
评论数 : 0
所有用户
所有用户
默认排序
默认排序
最近
早期
目前还没有评论。



通知
规格
数量
单价 (¥)
小计 1 (¥)
小计 2:
¥

规格
数量
单价 (¥)



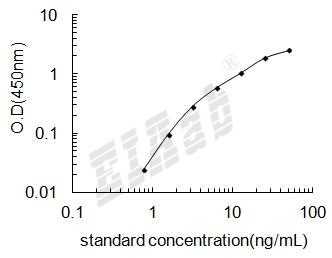
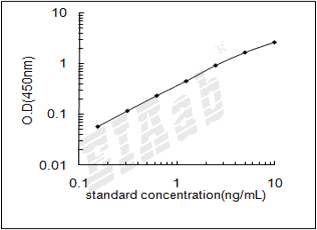
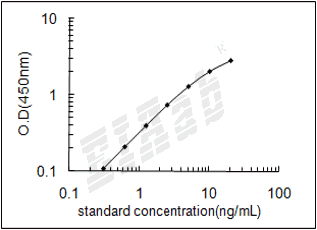
 验证序列:
验证序列:




 折扣:
折扣: