HSPA8 (基因名), Heat shock cognate 71 kDa protein (蛋白名), hsp7c_human.
产品名称:
Human HSPA8/ Heat shock cognate 71 kDa protein CLIA Kit
热休克蛋白质71kDa
货号:
U1094h
商标:
EIAab®
监管等级:
别名:
Heat shock 70 kDa protein 8, Lipopolysaccharide-associated protein 1, LAP-1, HSC70, HSP73, HSPA10
检测方法:
CLIA
实验类型:
Sandwich
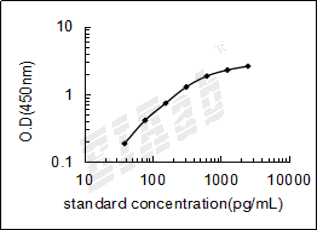
检测范围:
39-2500pg/mL
特异性:
Natural and recombinant human Heat shock cognate 71 kDa protein
样品类型:
Serum, plasma, tissue homogenates, cell culture supernates and other biological fluids
样品数据:
登录.
研究领域:
Epigenetics
通用注释
亚单元:
Identified in a IGF2BP1-dependent mRNP granule complex containing untranslated mRNAs. Interacts with PACRG. Interacts with HSPH1/HSP105. Interacts with IRAK1BP1 and BAG1. Interacts with DNAJC7. Interacts with DNAJB12 (via J domain) (PubMed:21150129, PubMed:21148293, PubMed:24732912, PubMed:27916661). Interacts with DNAJB14 (via J domain) (PubMed:23018488, PubMed:24732912, PubMed:27916661). Interacts (via C-terminus) with the E3 ligase CHIP forming a 210 kDa complex of one CHIP and two HSPA8 molecules. Interacts with CITED1 (via N-terminus); the interaction suppresses the association of CITED1 to p300/CBP and Smad-mediated transcription transactivation. Component of the PRP19-CDC5L splicing complex composed of a core complex comprising a homotetramer of PRPF19, CDC5L, PLRG1 and BCAS2, and at least three less stably associated proteins CTNNBL1, CWC15 and HSPA8. Interacts with SV40 VP1. Interacts with TRIM5. Part of a complex composed at least of ASCL2, EMSY, HCFC1, HSPA8, CCAR2, MATR3, MKI67, RBBP5, TUBB2A, WDR5 and ZNF335; this complex may have a histone H3-specific methyltransferase activity. Interacts with METTL21A. Following LPS binding, may form a complex with CXCR4, GDF5 and HSP90AA1. Interacts with PARK2. Interacts with FOXP3. Interacts with DNAJC9 (via J domain) (PubMed:17182002). Interacts with MLLT11 (PubMed:24880125). Interacts with RNF207 (PubMed:25281747). Interacts with DNAJC21 (PubMed:27346687). Interacts with DNAJB2 (PubMed:15936278). Interacts with TTC1 (via TPR repeats) (PubMed:15708368). Interacts with SGTA (via TPR repeats) (By similarity). Interacts with HSF1 (via transactivation domain) (PubMed:9499401). Interacts with HOPX, HSP40 and HSP90 (PubMed:27708256). Interacts with STUB1 (PubMed:27708256). Interacts with BAG2 (PubMed:24318877). Interacts with BAG3 (PubMed:27474739, PubMed:24318877).
功能:
Molecular chaperone implicated in a wide variety of cellular processes, including protection of the proteome from stress, folding and transport of newly synthesized polypeptides, activation of proteolysis of misfolded proteins and the formation and dissociation of protein complexes. Plays a pivotal role in the protein quality control system, ensuring the correct folding of proteins, the re-folding of misfolded proteins and controlling the targeting of proteins for subsequent degradation (PubMed:21150129, PubMed:21148293, PubMed:24732912, PubMed:27916661, PubMed:23018488). This is achieved through cycles of ATP binding, ATP hydrolysis and ADP release, mediated by co-chaperones (PubMed:21150129, PubMed:21148293, PubMed:24732912, PubMed:27916661, PubMed:23018488). The co-chaperones have been shown to not only regulate different steps of the ATPase cycle of HSP70, but they also have an individual specificity such that one co-chaperone may promote folding of a substrate while another may promote degradation (PubMed:21150129, PubMed:21148293, PubMed:24732912, PubMed:27916661, PubMed:23018488). The affinity of HSP70 for polypeptides is regulated by its nucleotide bound state. In the ATP-bound form, it has a low affinity for substrate proteins. However, upon hydrolysis of the ATP to ADP, it undergoes a conformational change that increases its affinity for substrate proteins. HSP70 goes through repeated cycles of ATP hydrolysis and nucleotide exchange, which permits cycles of substrate binding and release. The HSP70-associated co-chaperones are of three types: J-domain co-chaperones HSP40s (stimulate ATPase hydrolysis by HSP70), the nucleotide exchange factors (NEF) such as BAG1/2/3 (facilitate conversion of HSP70 from the ADP-bound to the ATP-bound state thereby promoting substrate release), and the TPR domain chaperones such as HOPX and STUB1 (PubMed:24318877, PubMed:27474739, PubMed:24121476, PubMed:26865365). Acts as a repressor of transcriptional activation. Inhibits the transcriptional coactivator activity of CITED1 on Smad-mediated transcription. Component of the PRP19-CDC5L complex that forms an integral part of the spliceosome and is required for activating pre-mRNA splicing. May have a scaffolding role in the spliceosome assembly as it contacts all other components of the core complex. Binds bacterial lipopolysaccharide (LPS) and mediates LPS-induced inflammatory response, including TNF secretion by monocytes (PubMed:10722728, PubMed:11276205). Participates in the ER-associated degradation (ERAD) quality control pathway in conjunction with J domain-containing co-chaperones and the E3 ligase STUB1 (PubMed:23990462).
亚细胞位置:
Cytoplasm
Melanosome
Nucleus
Nucleolus
Cell membrane
Localized in cytoplasmic mRNP granules containing untranslated mRNAs. Translocates rapidly from the cytoplasm to the nuclei, and especially to the nucleoli, upon heat shock.
该产品尚未在任何出版物中被引用。
反馈墙
评论数 : 0
所有用户
所有用户
默认排序
默认排序
最近
早期
目前还没有评论。



通知
规格
数量
单价 (¥)
小计 1 (¥)
小计 2:
¥

规格
数量
单价 (¥)




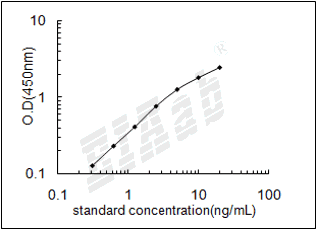
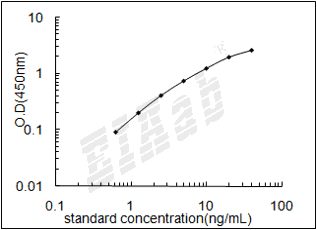

 验证序列:
验证序列:




 折扣:
折扣: